Snowflakes and a penny - OPOD
Snowflakes and a Penny: Unveiling the Beauty and Science Behind Snow Crystals
Snowflakes, those delicate ice crystals that grace our winter landscapes, have captivated the imaginations of people for centuries. Kenneth Libbrecht, a Professor of Physics at CalTech, not only collects these intricate formations but also grows his own. In one of his remarkable images, he compares the size of snowflakes to that of a US penny, showcasing the awe-inspiring beauty and complexity of these frozen wonders. The largest crystal in the image measures approximately 1 cm from tip to tip and holds the record as the largest individual snow crystal ever photographed. Let's delve deeper into the enchanting world of snowflakes and explore the science behind their formation.
Unveiling the Structure of Snow Crystals
At a molecular level, snowflakes are composed of ice crystals that reflect the underlying hexagonal structure of solid water. In a chemist's 'ball and stick' model, oxygen atoms are depicted as red, while hydrogen atoms occupy the spaces between them. The bonding of each hydrogen atom to two oxygen atoms creates an open and low-density structure in ice. This unique arrangement is responsible for the intriguing properties of ice, including its ability to float on water—a characteristic that has profound implications for life on Earth.
The Birth of a Snowflake
Snowflakes originate in cold clouds where tiny water droplets exist in a supercooled state, meaning they remain liquid below the freezing point. This metastable state sets the stage for the mesmerizing growth of snow crystals. When a droplet freezes, its vapor pressure becomes lower than that of the surrounding droplets. As a result, the crystal begins to grow, while the neighboring droplets evaporate to provide nourishment for its expansion.
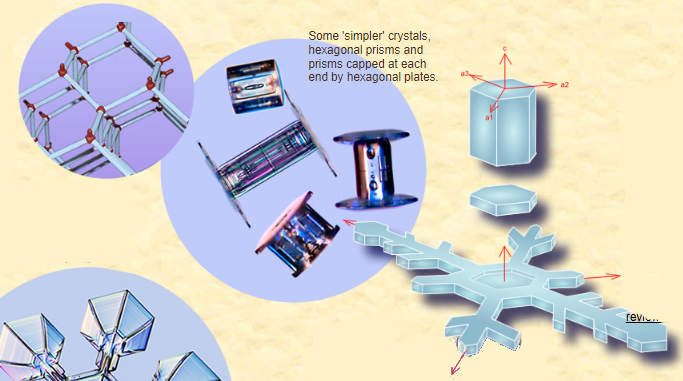
The Complexity of Snowflake Growth
The growth process of snowflakes is far from simple and regular. While hexagonal prisms occasionally reach a significant size, resulting in the formation of halos, more often, instabilities come into play. These instabilities lead to the growth of dendrites—branch-like structures that give snowflakes their intricate beauty, symmetry, and complexity. Dendrites themselves can even sprout their own dendrites, further enhancing the captivating patterns we observe in snowflakes.
Factors Influencing Snowflake Formation
Various factors influence the formation and appearance of snowflakes, such as humidity and temperature. These variables determine the intricate details and uniqueness of each crystal. For a comprehensive exploration of snow crystal physics, it is worthwhile to delve into Libbrecht's review on the subject. By understanding the interplay between these factors, scientists can gain valuable insights into the atmospheric conditions that shape snowflake morphology.
The Endless Variety of Snowflakes
Snowflakes exhibit an astonishing array of shapes and patterns, showcasing the boundless creativity of nature. From simple hexagonal prisms to elaborate dendritic structures, each snowflake is a unique masterpiece. The intricate branching patterns arise from the interplay between temperature, humidity, and other atmospheric conditions during crystal growth. This intricate dance between nature's elements ensures that no two snowflakes are ever identical—a testament to the complexity and diversity of our natural world.
Appreciating Snowflakes as Works of Art
Beyond their scientific significance, snowflakes also hold immense aesthetic appeal. The delicate lattices and intricate designs of these frozen masterpieces have inspired artists, poets, and dreamers throughout history. From children eagerly catching snowflakes on their tongues to photographers capturing their beauty on film, snowflakes have the power to ignite our sense of wonder and remind us of the enchantment that lies within the world around us.
Conclusion
Snowflakes, those ephemeral gems of winter, continue to captivate our imagination. Through the lens of science, we can uncover the intricate processes that give rise to their unique structures. From the molecular arrangement of ice crystals to the complex interplay of atmospheric conditions, each snowflake tells a story of its own. So, the next time you encounter a snowfall, take a moment to appreciate the delicate beauty of these frozen wonders and marvel at the secrets they hold.

Snow Crystals and a Penny
Kenneth Libbrecht, Professor of Physics at CalTech, collects snowflakes and grows his own. Here he compares them in size to a US Penny. “The largest crystal in that image is about 1 cm from tip to tip. I believe it still holds the record for the largest individual snow crystal ever photographed.”
See his books “The Secret Life of a Snowflake”, “Field Guide to Snowflakes” and more.
All images ©Kenneth Libbrecht, shown with permission
Ice crystals reflect the underlying hexagonal structure of solid water.
In this chemists' 'ball and stick' model, oxygen atoms are red.
Between each, but not equidistant, is a hydrogen atom. The bonding of each hydrogen to two oxygen atoms generates an open, low density, structure to ice. That's the reason ice floats on water. A factor to which we owe our existence.
Snowflakes start their life in a a cold cloud. Its small water droplets are below zero, supercooled. The supercooled state is metastable. If a droplet freezes then its vapour pressure is lower than that of the surrounding drops. The crystal grows and surrounding drops evaporate to feed the growth.
The growth is not simple and regular hexagonal prisms rarely reach a large size – when they do we see halos. More often, instabilities set in. The edges of facets grow more rapidly forming dendrites. Dendrites grow their own dendrites to give the beauty, symmetry and intricacy of a snow flake.
What happens depends on many factors like humidity and temperature. For more about it see Libbrecht’s review of snow crystal physics.

Note: this article has been automatically converted from the old site and may not appear as intended. You can find the original article here.
Reference Atmospheric Optics
If you use any of the definitions, information, or data presented on Atmospheric Optics, please copy the link or reference below to properly credit us as the reference source. Thank you!
-
<a href="https://atoptics.co.uk/blog/snowflakes-and-a-penny-opod/">Snowflakes and a penny - OPOD</a>
-
"Snowflakes and a penny - OPOD". Atmospheric Optics. Accessed on April 26, 2024. https://atoptics.co.uk/blog/snowflakes-and-a-penny-opod/.
-
"Snowflakes and a penny - OPOD". Atmospheric Optics, https://atoptics.co.uk/blog/snowflakes-and-a-penny-opod/. Accessed 26 April, 2024
-
Snowflakes and a penny - OPOD. Atmospheric Optics. Retrieved from https://atoptics.co.uk/blog/snowflakes-and-a-penny-opod/.