OPOD - Frost, hexagons and life
OPOD - Frost, Hexagons, and Life: A Closer Look at the Intricate Beauty of Ice Crystals
Ice crystals have long captivated our imagination with their delicate and intricate structures. From the simple columns and plates that create halos to the mesmerizing patterns of frost and snowflakes, these crystals exhibit a remarkable hexagonal form. This hexagonal shape is a direct reflection of the molecular structure of ice itself, which is strongly hexagonal.
At a molecular level, ice's hexagonal arrangement arises from the highly directional bonding between oxygen and hydrogen atoms, along with additional "hydrogen bonding." This unique arrangement produces an open and low-density crystal lattice, making it the most stable form of ice at everyday temperatures and pressures. The presence of hydrogen bonding, an electrostatic interaction between hydrogen and oxygen atoms, contributes to this unusual structure.
The consequences of ice's low density are far-reaching. Unlike most solids that sink in their liquid form, ice floats on water. This phenomenon has profound implications for our planet. If ice behaved differently and sank, Earth would be a vastly different place. Shallow bodies of water would freeze completely in winter, posing significant challenges for aquatic life. Moreover, the overall energy balance of the planet would be altered, potentially rendering life as we know it non-existent.
The hexagonal structure of ice not only impacts the existence of life but also influences the appearance of atmospheric phenomena such as halos. These captivating optical displays are formed by the interaction of light with ice crystals suspended in the atmosphere. The open hexagonal structure of ice plays a crucial role in shaping the intricate patterns observed in various types of halos. Without the hexagonal arrangement, these phenomena would take on markedly different forms, altering our perception of the sky.
To truly appreciate the beauty and complexity of ice crystals, one must witness them firsthand. The phenomenon of frost growth provides a stunning example of the hexagonal shape and layered structure of ice crystals. When exposed to freezing temperatures, water vapor in the air condenses directly onto surfaces, forming intricate frost patterns. These delicate ice crystals often exhibit clear hexagonal shapes, visually demonstrating the molecular structure of ice.
Photographers like Sandy Robertson have managed to capture the enchanting allure of frost growth through their lens. In natural light, the intricate hexagonal ice crystals shine, creating a captivating spectacle against the backdrop of a blue sky. Sandy's photo, taken using a reversed lens attached to the main lens, highlights the accessibility of macro photography and its ability to capture the intricate details of these frozen wonders.
The study of atmospheric optics continues to unravel the mysteries of natural phenomena, shedding light on the intricate beauty that surrounds us. By delving into the molecular structure of ice and its hexagonal arrangement, we gain a deeper understanding of the fundamental building blocks that give rise to the wonders of nature. From frost patterns on a shed roof to the ethereal beauty of halos in the sky, the hexagonal structure of ice reveals itself as a key player in shaping our world and enriching our lives.
In conclusion, the study of frost growth, hexagons, and ice crystals offers us a glimpse into the extraordinary beauty and intricacy of nature. The hexagonal structure of ice, driven by its molecular arrangement and hydrogen bonding, gives rise to captivating phenomena like frost patterns and halos. By appreciating these natural wonders, we gain a deeper appreciation for the fundamental forces that shape our world and the delicate balance necessary for life to thrive. So next time you encounter frost on a cold winter morning or gaze up at the sky adorned with halos, take a moment to marvel at the remarkable hexagonal beauty that surrounds us.
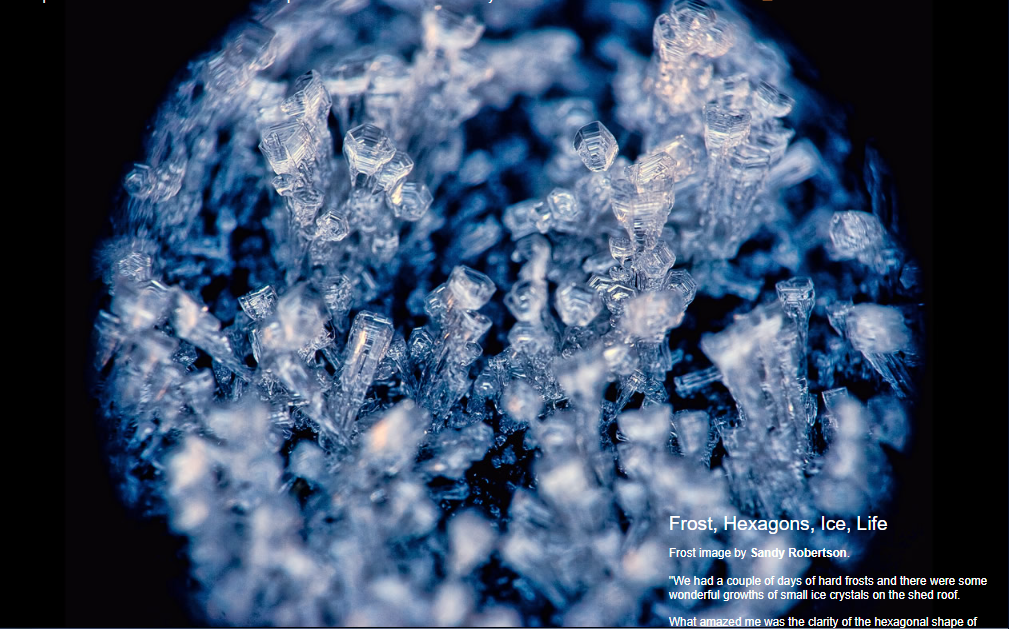
Frost, Hexagons, Ice, Life
Frost image by Sandy Robertson.
"We had a couple of days of hard frosts and there were some wonderful growths of small ice crystals on the shed roof.
What amazed me was the clarity of the hexagonal shape of some of the crystals and the way that they built up in layers (stacked plates). This really brought home to me how water can form hexagonal crystals.
I always find that however much one hears and 'understands' the theory, seeing a phenomenon in the flesh so to speak can add an extra level of appreciation........ and apart from that, I thought they looked really pretty in their own right!
The photo was taken under natural light (shadow, but blue sky) using a reversed lens (50mm) attached to the front of the main lens (18-55mm). It is a low cost way to get macro capability.
©Sandy Robertson, shown with permission
Ice crystals, from simple columns and plates that make halos to the intricate structures of frost and snowflakes, are all hexagonal in form. Their six-sidedness is a direct manifestation of ice�s molecular structure for that too is strongly hexagonal.
Ice�s hexagonal arrangement at a molecular level is a consequence of highly directional bonding between oxygen and hydrogen atoms combined with additional �hydrogen bonding�. The hydrogen bonding � an extra electrostatic interaction between an already bonded hydrogen atom and the oxygen of an adjacent water molecule � helps produce an uncommonly open and low density crystal lattice as the most stable ice form at everyday temperatures and pressures.
The low density has far reaching consequences. Ice is almost unique in that it floats on its own liquid � water. Nearly all other solids sink in their liquid form.
Earth would be quite different if ice did the same. Floating ice prevents shallow water from freezing completely in winter making aquatic life difficult or impossible. Worse, the overall energy balance of the planet would be different and life as we know it probably nonexistent.
Plus halos would have rather different shapes!
Two sights that we would not see but for the open hexagonal structure of ice.
Ice halos (Tom Faber) and icebergs (Les Cowley).
Ball and stick model of the molecular structure of ice. Red balls are oxygen atoms. Hydrogen atoms are between them along the white sticks but are not equidistant. Two hydrogen atoms are bonded strongly (covalently) to one oxygen atom and more loosely via 'hydrogen bonding' to an adjacent oxygen. The result is an unusually open, hexagonal, structure..

Note: this article has been automatically converted from the old site and may not appear as intended. You can find the original article here.
Reference Atmospheric Optics
If you use any of the definitions, information, or data presented on Atmospheric Optics, please copy the link or reference below to properly credit us as the reference source. Thank you!
-
<a href="https://atoptics.co.uk/blog/opod-frost-hexagons-and-life/">OPOD - Frost, hexagons and life</a>
-
"OPOD - Frost, hexagons and life". Atmospheric Optics. Accessed on April 19, 2024. https://atoptics.co.uk/blog/opod-frost-hexagons-and-life/.
-
"OPOD - Frost, hexagons and life". Atmospheric Optics, https://atoptics.co.uk/blog/opod-frost-hexagons-and-life/. Accessed 19 April, 2024
-
OPOD - Frost, hexagons and life. Atmospheric Optics. Retrieved from https://atoptics.co.uk/blog/opod-frost-hexagons-and-life/.