OPOD - Soap Film Colours
OPOD - Soap Film Colours: A Closer Look at the Beauty and Science Behind Soap Films
Soap films, those mesmerizing and iridescent bubbles, have captured the attention of many with their captivating colors and delicate structures. But what exactly makes these films so enchanting? In this article, we will dive deeper into the world of soap films and explore the science behind their vibrant hues.
The Magic of Soap Films
When we add soap or detergent to water, something magical happens. The water, which would normally sit placidly in a bowl or beaker, transforms into a world of airbourne bubbles filled with iridescent colors. These foams and bubbles possess intricate structures that continue to fascinate scientists and enthusiasts alike. With a bit of care, it is even possible to create large single-sheet films of remarkable delicacy and beauty.
The Role of Detergent Molecules
The key to water's behavioral shift lies in the presence of a single layer of detergent molecules. Water molecules naturally attract one another, creating a strong surface tension that minimizes the surface area. This surface tension is too strong for stable bubbles or single films to exist. However, when detergents are introduced, they significantly reduce the tension, allowing water to form thin films.
Detergent and soap molecules have polar groups on one end that attract water, while their hydrocarbon tails repel water but attract oils and greases. As a result, these molecules migrate to the water's surface, with their polar heads submerged in the water and their hydrocarbon tails protruding outwards. The soap films themselves consist of a sandwich of water molecules bordered by these ultra-thin detergent films. Additionally, the outer layers of detergent also help reduce evaporation, prolonging the lifespan of bubbles and films.
Understanding the Colors
The mesmerizing colors seen in soap films arise from the interference between light waves reflected from the front and back surfaces of the film. For example, consider the red waves on the far left. Some incident waves are directly reflected from the front surface, resulting in a 180° phase change. Other light enters the film and is internally reflected from the rear surface. When these two waves combine, they may either reinforce or cancel each other, depending on their phase relationship. In the case of the red waves, they are in phase, creating a visible red reflection in that direction.
The specific colors produced by soap films depend on the viewing angle and, most importantly, the thickness of the film. As the film drains and thins over time, the colors can change dynamically. Some colors with sharp boundaries may be the result of multi-layer films sliding over each other, creating an ever-changing display of hues.
The Mystery of Black
Among the stunning colors of soap films, black holds a special significance. When a film becomes dangerously thin, gravity causes it to drain downwards and become thinner at the top. This thinning process leads to an almost complete absence of phase difference between the waves reflected from the front and back surfaces. However, reflection from the front surface still produces a 180° phase shift. As a result, all wavelengths of light experience complete destructive interference, rendering the film black. Black in soap films is not a hole but rather a sign that the end is near, as the film or bubble is on the verge of collapse.
Exploring Soap Films Through Photography
Photographer Tim Durham has embarked on an extensive project to capture the beauty of single soap films through his lens. His photographs showcase the intricate patterns and vibrant colors that emerge from these delicate structures. The images, which span an area of 150 to 300 mm, aim to present the soap film interference colors as true to life as possible, without any enhancement or manipulation.
Conclusion
Soap films are not only visually captivating but also offer insights into the fascinating behavior of liquids and light. Through the addition of detergents, water transforms into delicate films that reflect a spectrum of colors through the interference of light waves. The ever-changing nature of soap films and their vibrant hues continue to inspire researchers, photographers, and enthusiasts alike. So next time you encounter a soap bubble or film, take a moment to appreciate the intricate beauty and the science behind its mesmerizing colors.

Soap Films
Soap film interference colours captured by Tim Durham (soap films, photography).
Tim has made an extensive project of photographing single soap films.
His films hang vertical and the area covered in these images was between 150 and 300 mm across.
The colours are as true to life as possible. There has been no enhancement or manipulation.
All images ©Tim Durham, shown with permission

Add some soap or detergent to water and we enter a new world.
Water, instead of sitting placidly in bowl or beaker, leaps out into airbourne bubbles of iridescent hues.
It turns into foams of incredible structure that we still do not fully understand and which refuse to quickly leave the bath.
And with some care we can make large single sheet films, as here, of great delicacy and beauty.

The key to water's behavioural shift is a single layer of detergent molecules.
Water molecules attract one another strongly. The net effect near its surface is to give it a 'surface tension' that acts to minimise the surface area. Water's surface tension is too strong for stable bubbles or single films to exist. Detergents considerably reduce the tension permitting water to form thin films.
Detergent and soap molecules have one end that is water attracting - they have heads with 'polar groups'. Their other end is a hydrocarbon tail that is water hating but attractive to oils and greases. Consequently the detergent molecules migrate to the water surface where the polar heads sit in the water and the hydrocarbon tails protrude out.
The soap films themselves are much thicker – they are a sandwich of water molecules edged by these ultra thin detergent films. The outer detergent layers also reduce evaporation, that enemy of long lasting bubbles and dedicated bubble blowers.
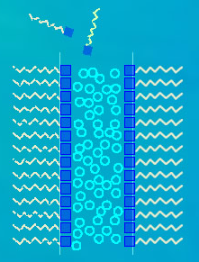
Detergent molecules cluster at the water film surface with their oleophilic water hating ends outwards and hydrophilic ends inwards..

The iridescent colours arise from interference between light waves reflected from the front and back film surfaces. Take the red waves at far left. Some of the incident waves are directly reflected from the front. Note that there is a 180° phase change when this happens. Some light enters the film and is internally reflected from the rear surface. The two waves combine. In the example at left the two waves are in phase and a red reflection would be seen in that direction.
Compare that with the shorter wavelength blue waves at near left. The two outgoing waves are out of phase and cancel each other. No blue would be seen.
The particular colours produced depend on viewing angle and most importantly, on the film thickness.

No two films are the same. They are ever changing as they drain and thin. Some of the colours with sharp boundaries can be the result of multi-layer films sliding over each other.
Black! Not a hole but a sign that the end is near. The film or bubble will soon collapse. Gravity has caused the vertical film to drain downwards and become thinner at the top. The black indicates that the film has become dangerously thin.
When the film is this thin there is almost no phase difference between the waves reflected from the front and back surfaces. BUT reflection from the front surface produces a 180° phase shift. As a result the two waves are completely out of phase at all wavelengths. The film becomes black.

Very thin film - Reflections from the front and back are out of phase and the film appears black.
Note: this article has been automatically converted from the old site and may not appear as intended. You can find the original article here.
Reference Atmospheric Optics
If you use any of the definitions, information, or data presented on Atmospheric Optics, please copy the link or reference below to properly credit us as the reference source. Thank you!
-
<a href="https://atoptics.co.uk/blog/opod-soap-film-colours/">OPOD - Soap Film Colours</a>
-
"OPOD - Soap Film Colours". Atmospheric Optics. Accessed on April 24, 2024. https://atoptics.co.uk/blog/opod-soap-film-colours/.
-
"OPOD - Soap Film Colours". Atmospheric Optics, https://atoptics.co.uk/blog/opod-soap-film-colours/. Accessed 24 April, 2024
-
OPOD - Soap Film Colours. Atmospheric Optics. Retrieved from https://atoptics.co.uk/blog/opod-soap-film-colours/.