Atomic fogbows and molecular beams
Atomic Fogbows and Molecular Beams: Unveiling the Intricacies of Atom Collisions
When we think of fogbows, we envision the ethereal arcs of light that grace misty skies. However, there is another type of fogbow that exists on a much smaller scale, one that is generated by colliding potassium atoms in a high vacuum apparatus. In this microscopic world, atoms behave like waves, exhibiting quantum mechanical properties that give rise to intriguing interference patterns. While the resemblance to a fogbow is not purely coincidental, it is important to note that only some aspects of the physics involved are similar.
In order to gain a deeper understanding of atom collisions, scientists employ various techniques to create controlled conditions for these interactions. One such method is through crossed molecular beams, where two narrow beams of atoms or molecules are directed towards each other in a high vacuum chamber. Within this controlled environment, the atoms undergo single collisions, allowing researchers to measure the way they scatter apart. By studying the scattering patterns, scientists can uncover intricate details about the forces between atoms and how they interact.
The scattering pattern produced by colliding atoms resembles a fogbow, but the underlying mechanisms are vastly different. To comprehend the intricacies of atom collisions, it is essential to explore the fundamental nature of atoms themselves. Unlike billiard balls that collide with a hard "clack" without any prior awareness of each other, atoms exhibit unique behavior due to the forces at play.
At large distances, atoms are attracted to each other. This attraction arises from fluctuating electron distributions in one atom inducing charge separation in the other, resulting in an attractive force. However, as the atoms approach each other, the attractive force switches to stronger and stronger repulsion. This repulsion stems from the positively charged nuclei of both atoms and a quantum effect known as Pauli exclusion repulsion, which prevents the collapse of neutron stars.
To predict an atom's path through this mixed attractive and repulsive force field, classical mechanics can be applied. However, classical mechanics fails to accurately describe atom behavior at everyday thermal energies. Instead, quantum mechanics must be employed. In this quantum realm, atoms are represented as waves, and a plane wave crossing the central force field replaces the atom particle. As the plane wave scatters, it generates an outgoing spherical wave, akin to a probability wave. The classical atomic rainbow spike transforms into a series of oscillations reminiscent of the primary and inner supernumeraries observed in a fogbow.
These oscillations within the scattering pattern hold valuable information about the interaction between atoms. By analyzing the structure of the oscillations, scientists can determine the interatomic forces at play. Notably, not all interatomic forces or collision energies result in the formation of a "rainbow" within the scattering pattern. Different forces and energies give rise to distinct scattering patterns, each providing unique insights into the underlying physics.
While theoretical predictions provide valuable information, experimental studies are crucial for confirming these predictions and uncovering further details about atom collisions. However, conducting experiments in this realm is no easy feat. The source atoms must be collimated into strongly directional beams using slits, and these beams are then filtered to pass only narrow velocity ranges. Additional properties such as mass, rotational state, or electronic spin may also be selected. Despite these meticulous preparations, the resulting beams are often weak, and only a few collisions occur when they intersect. Detecting and measuring individual atoms or molecules require further filtering and sophisticated techniques such as counting emitted light during collisions.
Although experimental observations may not always capture the full extent of theoretical predictions, they still offer valuable insights into the forces between atoms and molecules. These observations provide detailed information about collision dynamics, energy exchange, and even the birth of new molecules through chemical reactions. Molecular beam research delves into the intricacies of atom collisions, shedding light on the fundamental processes occurring at the microscopic level.
In conclusion, the study of atomic fogbows and molecular beams allows scientists to explore the complex world of atom collisions. Through crossed molecular beams and meticulous experimental techniques, researchers can uncover intricate details about the forces between atoms, the way they interact, and the formation of scattering patterns resembling fogbows. By combining theoretical predictions with experimental observations, scientists continue to unravel the mysteries of atom collisions and deepen our understanding of the fundamental building blocks of our universe.
Atomic Fogbow
Not the familiar fogbow that graces water droplet laden skies. Instead a scattering pattern generated by potassium atoms colliding in a high vacuum apparatus. The atoms behave like waves that interfere. Waves of quantum mechanical probability if you like. The resemblance to a fogbow is more than coincidental for some, not all, of the physics is similar.
All images ©Les Cowley
Atoms and molecules collide incessantly in the air. But to glean knowledge from collisions they need to be made (footnote 1) under far more controlled conditions.
One way is via crossed molecular beams. Two narrow beams of atoms or molecules are directed at each other in a high vacuum chamber (2). The atoms make a single collision and we measure the way they scatter apart.
The scattering pattern above is a theoretical prediction for atoms hitting each other at 1 km/s. That is an ordinary thermal energy. Measurements of scattering patterns reveal fine details of the forces between atoms and how they interact.
Fogbows and atomic scattering
A - A fogbow made by 10 micron dia. water droplets. It has prominent supernumeraries and a central glory.
B - The same fogbow in monochromatic light. Mie predictions have strong angular fluctuations shifting rapidly as the wavelength changes.
C - A scattering pattern from colliding atoms
Atomic fogbows and rainbows
Billiard balls strike with a hard 'clack'. Before that they are quite unaware of each other.
Atoms are different. At large distances they attract each another. Fluctuating electron distributions in one induce charge separation in the other - result, attraction. As they approach the attraction switches to stronger and stronger repulsion. The repulsion is from the two positively charged nuclei plus a quantum effect – Pauli exclusion repulsion. Pauli exclusion is the same force that stops neutron stars collapsing.
We might try classical mechanics to predict an atom's path though this mixed attractive/repulsive force field (3). Some trajectories are at right. The path starting at ‘a’ is hardly affected. As the atom skims closer to the force centre it deflects more. Inside ‘b’, repulsive forces dominate. At 'c' the atom deflects strongly in the opposite direction.
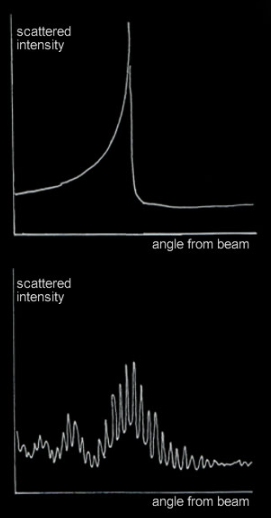
At ‘b’ the deflection turns around and paths cluster there - Just as light rays do to form a rainbow! Classically, we would see a large spike in the scattered atom intensity at a certain angle – an atomic rainbow.
What actually happens is more interesting. At everyday thermal energies, classical mechanics doesn't work and we must instead use quantum mechanics.
A plane wave crossing the central force field replaces the atom particle. The plane wave scatters to make an outgoing spherical wave (4). A probability wave perhaps. The classical atomic rainbow spike transforms into a series of oscillations analogous to a fogbow’s primary and inner supernumeraries.
Those oscillations are a rich source of information about the interaction.
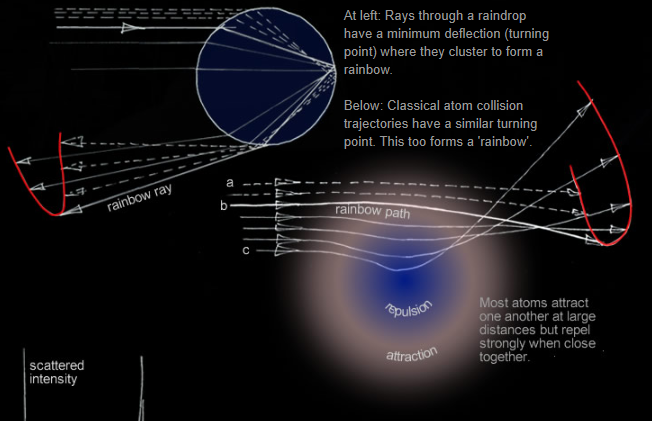
At left: Rays through a raindrop have a minimum deflection (turning point) where they cluster to form a rainbow.
Below: Classical atom collision trajectories have a similar turning point. This too forms a 'rainbow'.
A rainbow spike for elastic atom collisions predicted by classical mechanics. Like an optical rainbow, the angles within the rainbow are bright and the outside dark.
Not all interatomic forces or collision energies give a rainbow.
An equivalent quantum mechanics prediction. The atoms behave as waves. The classical rainbow spike is replaced by broader peaks, inner supernumeraries and a superimposed higher angular frequency oscillatory structure.
Experimental measurements on the structure enable determination of the interatomic forces.
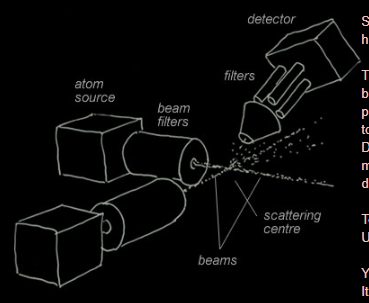
Meanwhile, back in the lab
Someone once said that molecular beam research was like peering hopefully (or hopelessly!) at the ragged edge of invisibility.
The source atoms must be collimated by slits into strongly directional beams. The beams are filtered to pass only narrow velocity ranges. Other properties like mass, rotational state or electronic spin might be selected too. All that gives weak final beams and few collisions where they cross. Detectors need further filtering and have to count individual atoms or molecules. Alternatively, some methods detect instead the light emitted during the collisions.
To detect the full detail of theoretical predictions would be marvelous. Usually only major features might be discerned.
Yet what is seen tells directly of the forces between atoms and molecules. It tells how they collide and exchange energy. It reveals intimate step by step details of how some react to birth new molecules.
For fascinating accounts of early research on molecular beams and chemical dynamics see the Nobel lectures of Dudley Herschbach and John Polanyi

The small print
-
Atoms in the air or gases collide furiously at a whole range of speeds and directions. Properties that depend on collisions like viscosity or chemical reaction rates are huge averages over this range and measuring them tell us little about the individual elementary collisions and fundamental processes occurring.
-
The best possible vacuum is needed to avoid secondary collisions with the chamber's residual background gas. Not easily achieved and stages of differential pumping are employed.
-
This text talks mostly about 'elastic' collisions, i.e. those where there is no change in total translational energy. Many other collision interactions occur where there are changes in translational, electronic, vibrational and rotational energy not to mention spin and exchange of atoms between the colliding groups - chemical reaction.
The 'two body' problem of colliding atoms can be exactly replaced by that of a single body of reduced mass passing across a potential energy surface V(r). V(r) is that existing between the two original atoms with r the interatomic distance. The trajectories shown here are in that spirit. Gravitational attraction is similarly treated but gravity falls off slowly at the inverse square of distance. Atomic forces are shorter range and vary as the inverse sixth to twelfth power.
- Briefly, radial wave differential equations are solved for up to 1000 values of the collision angular momentum quantum number 'L'. For each 'L' the phase shift is derived for waves with and without the potential V(r) acting. All the collision information is in the phase shifts. Scattering cross sections for a range of angles are then computed using a 'partial wave' method first derived by Lord Rayleigh for the scattering of sound waves through balloons of different gases!
Note: this article has been automatically converted from the old site and may not appear as intended. You can find the original article here.
Reference Atmospheric Optics
If you use any of the definitions, information, or data presented on Atmospheric Optics, please copy the link or reference below to properly credit us as the reference source. Thank you!
-
<a href="https://atoptics.co.uk/blog/atomic-fogbows-and-molecular-beams/">Atomic fogbows and molecular beams </a>
-
"Atomic fogbows and molecular beams ". Atmospheric Optics. Accessed on April 20, 2024. https://atoptics.co.uk/blog/atomic-fogbows-and-molecular-beams/.
-
"Atomic fogbows and molecular beams ". Atmospheric Optics, https://atoptics.co.uk/blog/atomic-fogbows-and-molecular-beams/. Accessed 20 April, 2024
-
Atomic fogbows and molecular beams . Atmospheric Optics. Retrieved from https://atoptics.co.uk/blog/atomic-fogbows-and-molecular-beams/.